|
The definitions of molecular geometry and electronic geometry are different. A represents the central atom, each X represents an atom bonded to A (or bond pair), and each E represents a lone pair on the central atom.īelow is the table of molecular geometries, arranged for different electron pairs:Īre electron geometry and molecular geometry definitions the same? AXE MethodĪXE method is an efficient tabular idea to represent molecular geometries. Various techniques to determine molecular geometry include Raman spectroscopy, infrared spectroscopy, and microwave spectroscopy. It influences a substance’s properties such as its reactivity, color, polarity, magnetism, biological activity, and phase of matter. Molecular geometry includes geometrical parameters such as bond lengths, bond angles, and torsional angles that help determine the position of atoms as well as a molecule’s general shape. The shape of a molecule is determined by the bonded atom, although this does not mean the shape itself is unaffected by the presence (repulsion) of lone pair(s). The following table gives an idea of electronic geometry according to the number of electron pairs. Since electrons are always moving and their paths cannot be accurately figured, the arrangement of electrons is described in terms of electron density distribution.Įlectron geometry is determined by the number of electron pairs. 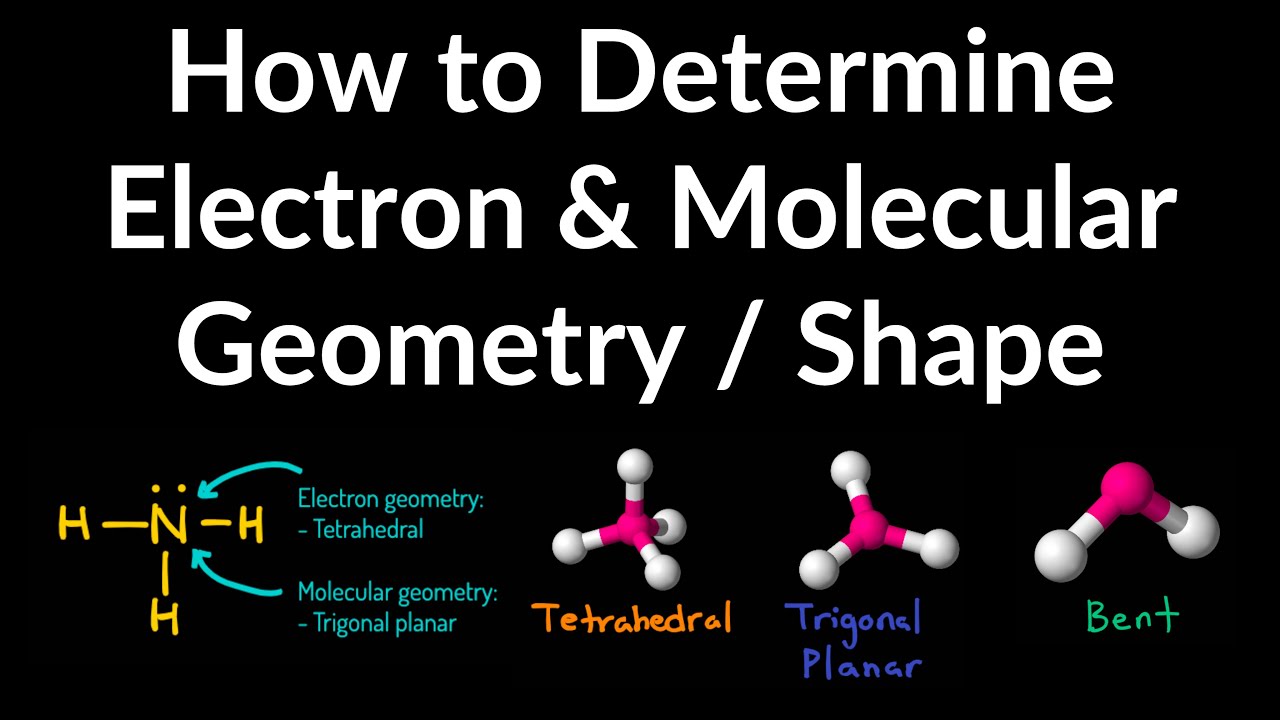
It considers the presence of both bond pair(s) and lone pair(s) of electrons in determining the shape. It excludes lone pairs in deciding the shape of a molecule, although repulsion from lone pair(s) is taken into account only in bond angles. Molecular Geometry is the arrangement of atoms in a molecule, normally relative to a single central atom.Įlectron Geometry is the arrangement of electron pairs around a central atom. Valence Shell Electron Pair Repulsion (VSEPR) Theoryĭifference between molecular geometry and electron geometry.Lewis Structure (Representation of Valence Electrons in a molecule).Its molecular geometry is trigonal pyramidal while its electron geometry is tetrahedral. With the central atom nitrogen having 5 valence electrons, it possesses 3 bond pairs and a lone pair of electrons. Its molecular, as well as electronic geometry, is tetrahedral.Īn example with differing molecular and electron geometries is that of ammonia, NH 3. An example is a methane molecule, CH 4 with 4 bond pairs and no lone pairs, all 4 of carbon’s valence electrons are bonded with hydrogen atoms. If all of the electron groups are bond pairs (no lone pairs), the molecular geometry and electron geometry are the same. This causes a slight decrease in bond angles (angles between bonds or bond pairs). Under the influence of a single nucleus, a lone pair offers more repulsion than a bond pair which is influenced by two nuclei. This repulsion causes the electron pairs around the central atom to arrange as far apart from each other as possible. 
It is well known that the electron pairs, being negatively charged, repel each other. 
Whereas, electron geometry is the 3D arrangement of electron pairs around a central atom, whether bonding or non-bonding.Ī lone (non-bonding) pair refers to a pair of valence electrons that are not shared with another atom in a covalent bond, while a bond pair is a pair of electrons present in a bond. Molecular geometry is described as the 3D arrangement of atoms in a molecule, normally relative to a single central atom. Geometry in chemistry refers to the shape of molecules in 3-Dimensional space.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
